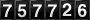
KD=C1C2cap K sub cap D equals the fraction with numerator cap C sub 1 and denominator cap C sub 2 end-fraction C1cap C sub 1 C2cap C sub 2
The Nernst Distribution Law simplifies the complex behavior of solutes in multi-phase systems. By establishing a predictable ratio of concentration, it allows chemists to manipulate chemical environments for extraction and analysis. Whether it is purifying a compound in a lab or determining how a medication will move through the human body, the Distribution Law remains an indispensable tool in modern science. distribution law
For the law to hold strictly true, several conditions must be met: KD=C1C2cap K sub cap D equals the fraction
The principle of a solute partitioning itself between a mobile phase and a stationary phase is the fundamental mechanism behind various chromatographic separation techniques. Conclusion For the law to hold strictly true, several
This is the most common application. It is used to remove a desired substance from a solution by shaking it with a second, immiscible solvent in which the substance is more soluble. This is a staple technique in organic chemistry and pharmaceutical manufacturing.
In pharmacology, the partition coefficient (often expressed as Log P) helps scientists predict how easily a drug can cross cell membranes, which are primarily composed of lipids.